 
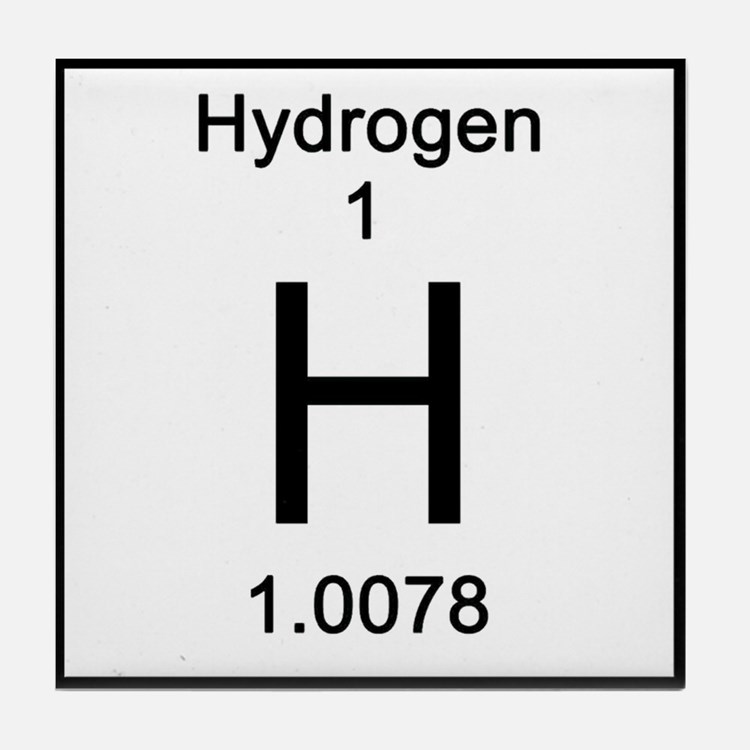
The atomic mass of an element is the weighted mass of all the naturally presented isotopes (on earth). Since most naturally occurring elements samples are mixtures of isotopes, it is useful to use an average weight of an element. “Blausen 0527 Hydrogen-2 Deuterium” Av BruceBlaus – Eget verk (CC BY 3.\) compares the three isotopes of hydrogen. “Blausen 0526 Hydrogen-1Atom” By BruceBlaus – Own work (CC BY 3.0) via Commons Wikimedia “Hydrogen – Element information, properties and uses | Periodic Table.” Royal Society of Chemistry – Advancing excellence in the chemical sciences. “Interesting Deuterium Facts.” ThoughtCo. However, in addition to being isotopes, there is a distinct difference between Deuterium and Hydrogen. Since Protium is the most abundant isotope of hydrogen, it is considered as normal hydrogen. Summaryĭeuterium is an isotope of hydrogen. Hydrogen: The abundance of Hydrogen is about 99%. Natural Abundanceĭeuterium: The abundance of Deuterium is about 0.015%. Hydrogen:Hydrogen can be represented as either H or 1 1H. Elemental Symbolĭeuterium: Deuterium can be represented as either D or 2 1H. Hydrogen:The atomic mass of Hydrogen is 1.00794 u. Atomic Massĭeuterium: The atomic mass of Deuterium is 2.014 u. Hydrogen: Hydrogen is a chemical element with the atomic number 1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
